The EcoRI restriction sites are still highlighted in the list and I also added the insertion site as a feature in the map. When you go to the graphic map of your plasmid, you get something like in the image below. It is also very useful to give your feature a unique color. Is it a gene, a primer, a binding site or something else. It is also possible to specify the feature type (green arrow). You can give a name to the feature you added (blue arrow). If you click on ‘New Feature’, while the sequence of your sequence is still highlighted in the main window, a new window opens like the one shown below. When you click on it a drop down menu opens. To annotate various components in the plasmid map you will find a button in the main toolbar, at the top of the screen, called ‘Features’. Which then looks something like in the image below. Once I have saved the sequence of the insert in a separate file, I can copy it and past it at the correct site in my vector. It will get messy, if you are trying to revert changes. If you make changes to your original vector, safe it as a separate file. Note: Do not forget to save your files individually. However, this might not be the ideal probe for in situ hybridization, since I have not included any of the UTRs or checked for alternate transcripts. To demonstrate my method, I have picked the cDNA sequence of the zebrafish gene shha. I got the sequence from the Ensembl database and copy-pasted it into a new ApE window, just like I did with the vector.

You can select and de-select an enzyme simply by clicking on it.įlanking the area, where our DNA fragment will be inserted are the EcoRI restriction sites. If you press this button, the program will show, where in your sequence the restriction sites are for the enzymes you have selected. In the toolbar below the list, where it says ‘Perform Action’, is a button called ‘Highlight’. Highlighted is EcoRI and it indeed just cuts twice in the vector. The numbers next to the name of the enzyme indicate, how many times the enzyme cuts in the construct. The toolbar contains an icon, which opens a list of all the common restriction enzymes (and some not so common ones) in a separate window. These are the only two EcoRI sites in the plasmid. The diagram tells us, that the insertion site is flanked by two EcoRI restriction sites. Going back to the vector information, we have to find out where our PCR fragment will be inserted. Now, if you press on the ‘Graphic Map’ icon (green arrow in image below) you will get a map of your circular plasmid.īut there is not much to see yet, since it is not annotated Since this is a plasmid, I set it to circular. The green arrow in the top right corner highlights a toggle switch between ‘Linear’ and ‘Circular’.
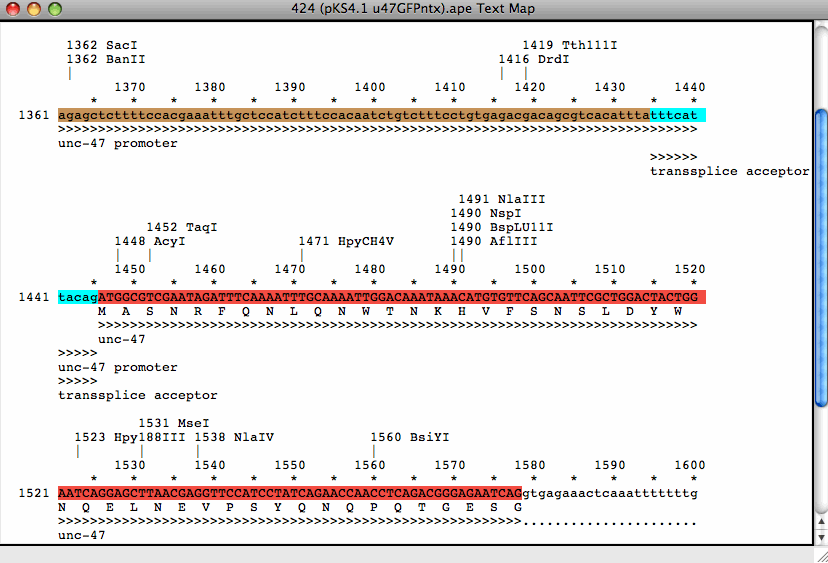
To make explaining a little easier, I will use the pCR Blunt II TOPO vector and the cDNA sequence of the zebrafish gene shha as an example.įirst I copied and pasted the sequence of the vector into the empty window of ApE. There is a main toolbar, in my case at the top of the screen, and there are icons in the window, where the sequence is pasted. Once you have installed the program, this is the kind of window you will get. The nice thing about ApE is, that it is intuitive and has all the tools and functions you need to design your cloning strategies. Below is a description of how I go about making my plasmid maps.
Ape dna editor paste rna sequence software#
To do so I use a freely available software called ‘A plasmid editor’ or ApE. Since I prefer to clone my riboprobes, rather than amplifying them every time from cDNA, I have to making in silico maps of the constructs first.
